|
12/27/2023 0 Comments Explain resonance in chemistry 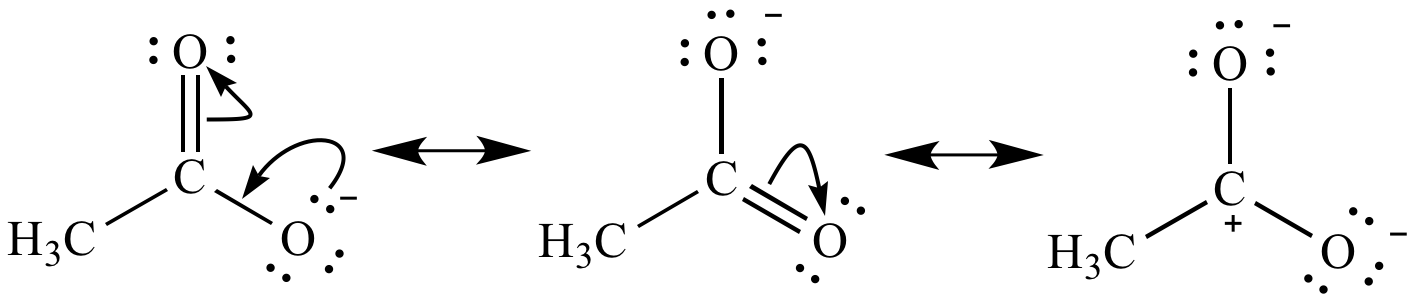
Resonance contributors for the same molecule all have the same chemical formula and same sigma framework, but the pi electrons will be distributed differently among the atoms. The electron density of these bonds is spread over the molecule, also known as the delocalization of electrons. Resonance structures are diagrammatic tools used predominately in organic chemistry to symbolize resonant bonds between atoms in molecules. Using laboratory procedures to measure the bond length of each bond, we do not find that one bond is shorter than the two others (remember, double bonds are shorter than single bonds), but instead that all bonds are of the same length somewhere between the length of typical double and single bonds. In the Lewis structures above, carbonate (CO 3 2-) has a resonance structure. Resonance stability plays a major role in organic chemistry due to resonant molecules' lower energy of formation, so students of organic chemistry should understand this effect and practice spotting molecules stabilized by resonant forms. Other aromatic molecules have a similar stability, which leads to an overall entropic preference for aromaticity (a subject that will be covered fully in a later chapter). This is the reason that benzene (C 6H 6) has a lower heat of formation than organic chemists would predict, not accounting for resonance. Resonance structures are stabilizing in molecules because they allow electrons to lengthen their wavelengths and thereby lower their energy. Resonance is an expression of this uncertainty, and is therefore the average of probable locations. Chemists are absolutely certain where electrons are located when one carbon bonds four hydrogens (methane), but it is less certain where precisely any given electron is located when six carbons bond six hydrogens in a ring structrue (benzene). What a dot structure is actually showing is where electrons almost certainly are located, therefore resonance structures indicate a split in those same probabilities. The true situation is that no one can say for certain exactly where any individual electron is at any specific moment, but rather electron location can be expressed as a probability only. Rather, the electrons are portrayed as if they were moving instead. The nuclei of the atoms are not moving when they are represented by resonance structure drawings. The result of all that complexity is simply this: molecules with resonance structures are treated as mixtures of their multiple forms, with a greater percentage of probability given to the most stable configurations. Changes in molecular shape occur so rapidly, and on such a tiny scale, that the actual physical locations of individual electrons cannot be precisely known (due to Heisenberg's Uncertainty Principle). The actual situation on the molecular scale is that each configuration of the molecule contributes a percentage to the possible configurations, resulting in a "blend" of the possible structures. Just as entropic principles cannot be applied to individual molecules, it is impossible to say whether or not any given individual molecule with a resonance structure is literally in one configuration or another. It is sometimes best to use analogies to introduce a topic, but then explain the differences and inevitable complications as further details on a complicated subject. In science, analogies can provide an aid to understanding, but analogies should not be taken too literally. Resonance is easily misunderstood in part because of the way certain chemistry textbooks attempt to explain the concept. 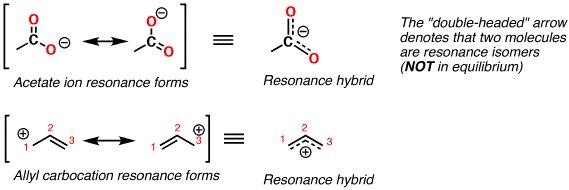
Resonance refers to structures that are not easily represented by a single electron dot structure but that are intermediates between two or more drawn structures. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
